|
12/13/2023 0 Comments Sf4 dipole moment 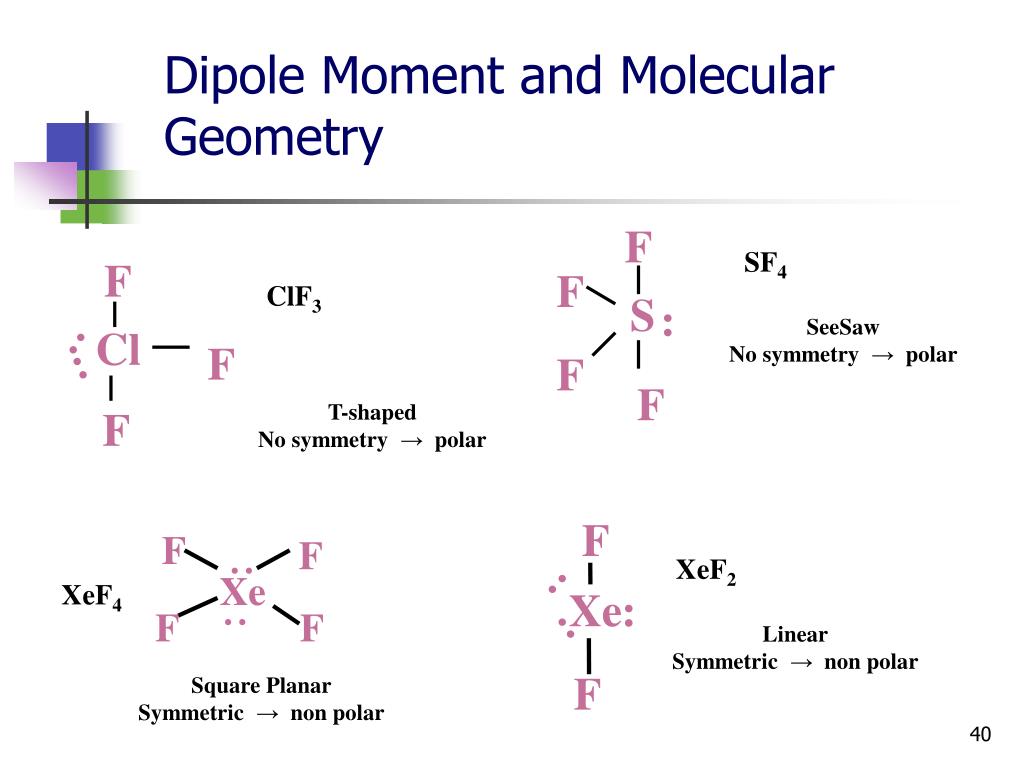
The instrument used for the measurements was a 5 kc Stark modulated spectrograph. The SF, lines were initially identified as those which disappeared when the sample was treated with water. The microwave lines arising from SOF2 are quite intense and even at 5% abundance they appeared stronger than the SF4 lines. The main impurity was SOF2 present in aboutġ D. The sample of SF4 was obtained through the courtesy of H. The microwave studies were undertaken to determine the detailed structure of SF. Which showed that SF4 has two inequivalent sets of fluorine atoms. This has been confirmed by recent nuclear magnetic resonance studies4,5 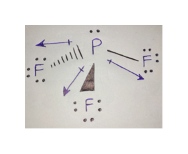
Previous structural information for SF, has come from the IR and Raman spectra3 which indicate a structure having C2v symmetry. Studies on the Raman spectrum, however, had given indication of the less symmetrical C2v structure. In later work on SeF, Bowen2 was unable to explain his electron diffraction patterns without assuming that both opposing F-Se-F angles were equal and that the four bonds were of equal length (D2d symmetry). The first measurement on this series of compounds was the electron diffraction study of TeC14.1 Here, assuming all Te-Cl bonds to be of equal length, it was found that the opposing Cl-Te-Cl angles were 170o☑5° and 93°☓°. 
6, 152 (1961).JĪLTHOUGH some conflicting evidence does exist, ft most structural determinations of the group VI tetrahalides have led to the conclusion that these molecules have C2v symmetry. The dipole moment was determined from the Stark effect to be 0.632☐.OO3 debye. The bond length for the nearly linear bonds is 1.646 A☐.OO3 A for the other pair it is 1.545 A☐.OO3 A. One FSF angle is 101☃3'☓0', the opposite FSF angle is 186★6'☓0'. The microwave spectrum and structure of sulfur tetrafluoride has been determined. Downloaded to IP:ĬHEMICAL PHYSICS VOLUME 36, NUMBER 5 MARCH 1, 1962ĭepartment of Chemistry, University of California, Berkeley, California Reuse of AIP content is subject to the terms at. This article is copyrighted as indicated in the article. Gwinn Citation: The Journal of Chemical Phys(1962) doi: 10.1063/1.1732702 View online: View Table of Contents: Published by the AIP Publishing Articles you may be interested in Microwave spectrum, structure, and dipole moment of 2,3,4,5tetracarbahexaborane(6) J. Gwinn Citation: The Journal of Chemical Physics 36, 1119 (1962) doi: 10.1063/1.1732702 View online: View Table of Contents: Published by the AIP Publishing Articles you may be interested in Microwave spectrum, structure, and dipole moment of 2,3,4,5tetracarbahexaborane(6) J. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
